Hydrogen is widely used in steel, metallurgy, chemical industry, medical, light industry, building materials, electronics and other fields. Methanol reforming technology to produce hydrogen has the advantages of low investment, no pollution, and easy operation. It has been widely used in all kinds of pure hydrogen plant.
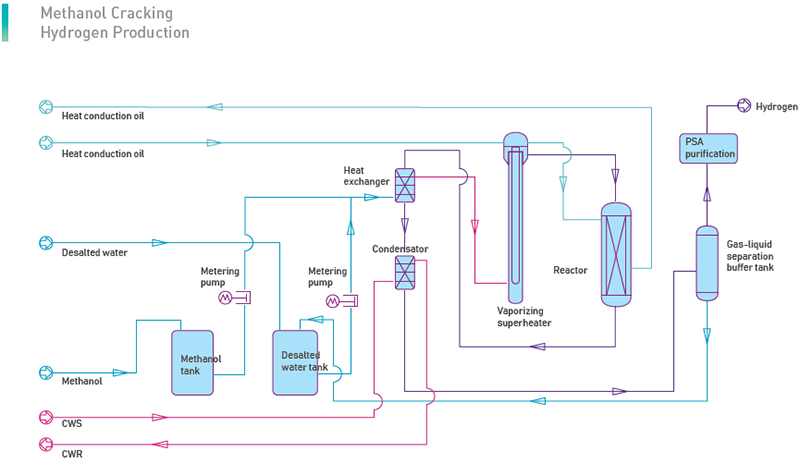
Mix methanol and water in a certain proportion, pressurize, heat, vaporize and overheat the mixture material to reaches a certain temperature and pressure, then in the presence of catalyst, methanol cracking reaction and CO shifting reaction perform at the same time, and generate a gas mixture with H2, CO2 and a small amount of residual CO.
The whole process is an endothermic process. The heat required for the reaction is supplied through the circulation of the heat conduction oil.
To save heat energy, the mixture gas generated in the reactor makes heat exchange with the material mixture liquid, then condenses, and is washed in the purification tower. The mixture liquid from the condensation and washing process is separated in the purification tower. The composition of this mixture liquid is mainly water and methanol. It is sent back to the raw material tank for recycling. The qualified cracking gas then is sent to the PSA unit.










 CO + 2H2 – 90.7kJ/mol
CO + 2H2 – 90.7kJ/mol











